Management & Organisational
Guidelines for Control of Cosmetic Products in Malaysia
What this infographic shows
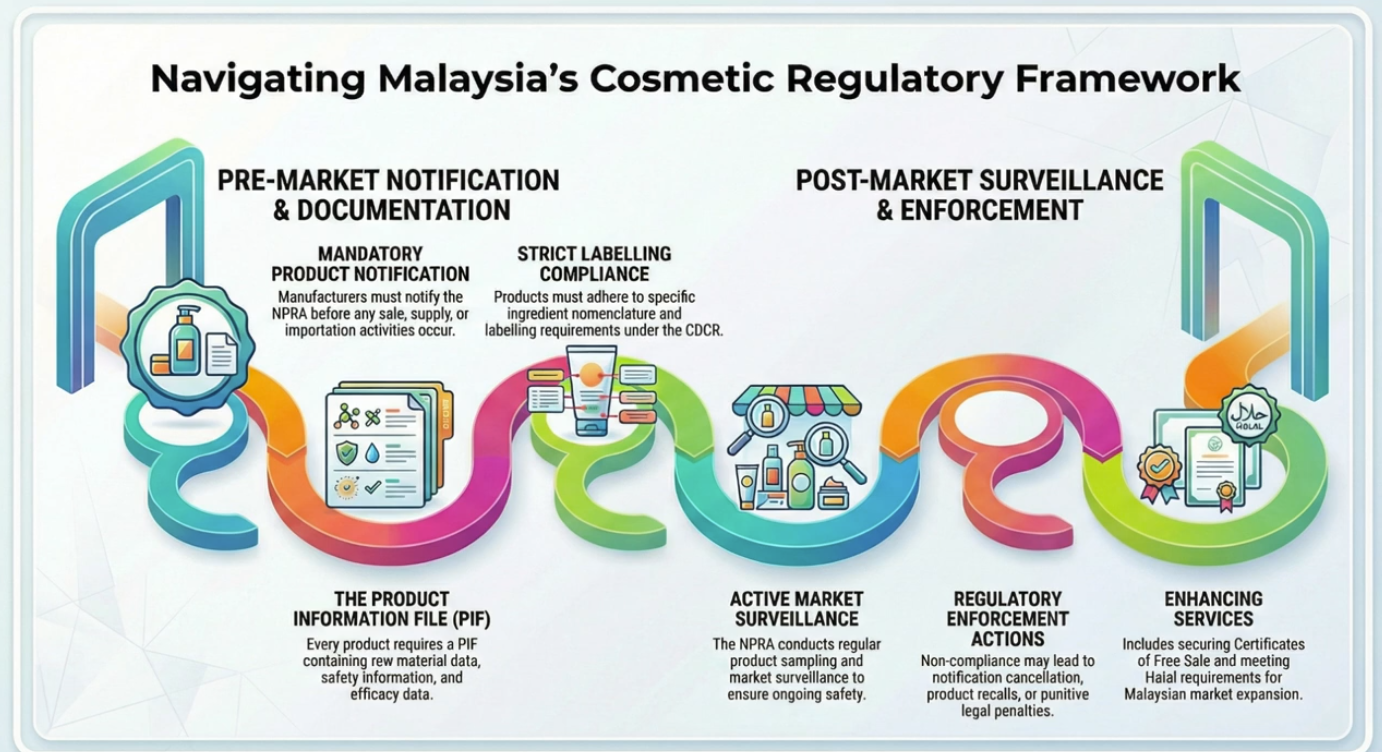
The Control of Cosmetic Products Guidelines has been produced to offer assistance to the
cosmetic industry in compliance with the provisions of the ASEAN Cosmetic Directive
(ACD). The course is for the manufacturers to understand that Cosmetic products in Malaysia
are regulated under the Control of Drugs and Cosmetic Regulations (CDCR) 1984, which
were promulgated under the Sale of Drugs Act 1952. Cosmetic products in Malaysia are
controlled through notification procedure starting from 1 st January 2008. Cosmetic
Notification Holder (CNH) who responsible for placing the cosmetic product in the market is
required to COMPLY with all requirements stated in the guideline and to make a declaration
upon notification to the Director of Pharmaceutical Services (DPS) through National
Pharmaceutical Regulatory Agency (NPRA). This training is to create awareness of the
Control of Drugs and Cosmetic Regulation (CDCR 1984), Regulation 18A (1) that is: No
person shall manufacture, sell, supply, import, possesses any cosmetics unless the cosmetic is
notified. It is an offence for anyone to conduct such activities without prior notification to the
DPS.
Want to explore this further?
This infographic highlights key insights connected to deeper learning and practical application. Discover programmes related to this topic or speak to us about how these insights can be applied in your organisation.
- ✔ Programmes aligned with this infographic
- ✔ Practical learning beyond the visual summary
- ✔ Facilitator-led and evidence-informed approaches
- ✔ Suitable for individuals & organisations